“The part 3 (trial) is presently ongoing. We’re dosing sufferers and we anticipate to finish the dosing as a part of the part 3 by February. After that we anticipate to compile the information and submit for Emergency Use Authorisation, make the request to the DCGI with our file,” firm’s CEO of APIs and Pharmaceutical Companies Deepak Sapra stated.
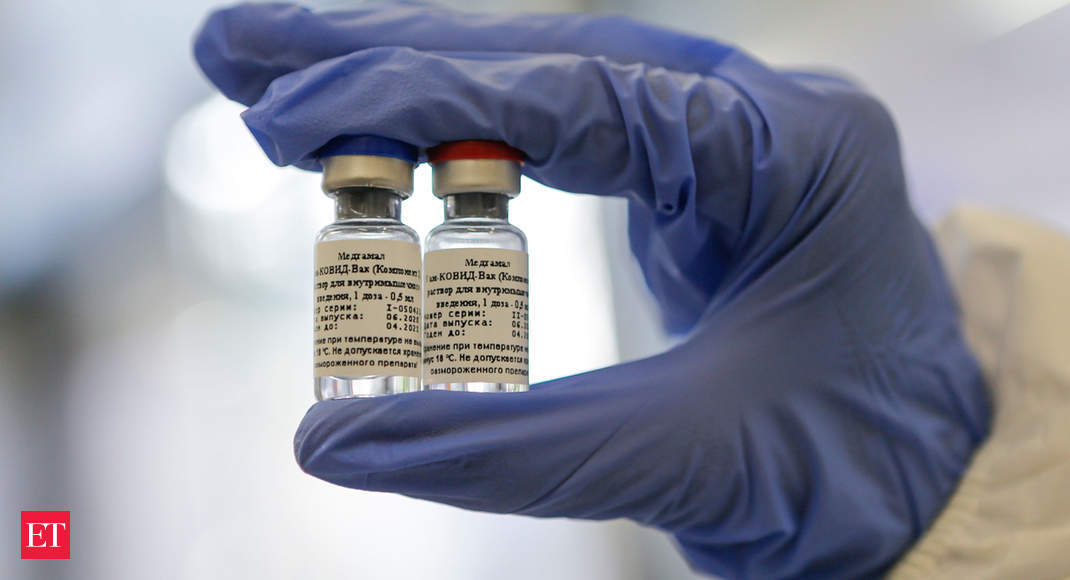
In September final, Dr. Reddy’s partnered with Russian Direct Funding Fund (RDIF) to conduct the scientific trials of the Sputnik V vaccine and for its distribution rights in India.
“And primarily based on the approval from the DCGI, we consider that we needs to be ready to launch the vaccine by means of a EUA within the month of March 2021 (in India), Sapra instructed a press convention right here.
Sputnik V, developed by the Gamaleya Nationwide Analysis Institute of Epidemiology and Microbiology, was registered by the Ministry of Well being of Russia and have become the worlds first registered vaccine in opposition to COVID-19 primarily based on the established human adenoviral vector platform.
The Centre has already given the nod for 2 COVID-19 vaccines — Covaxin of city-based Bharat Biotech and Covishield of Oxford, being manufactured by Serum Institute of India in Pune, that are being administered to frontline employees from January 16.