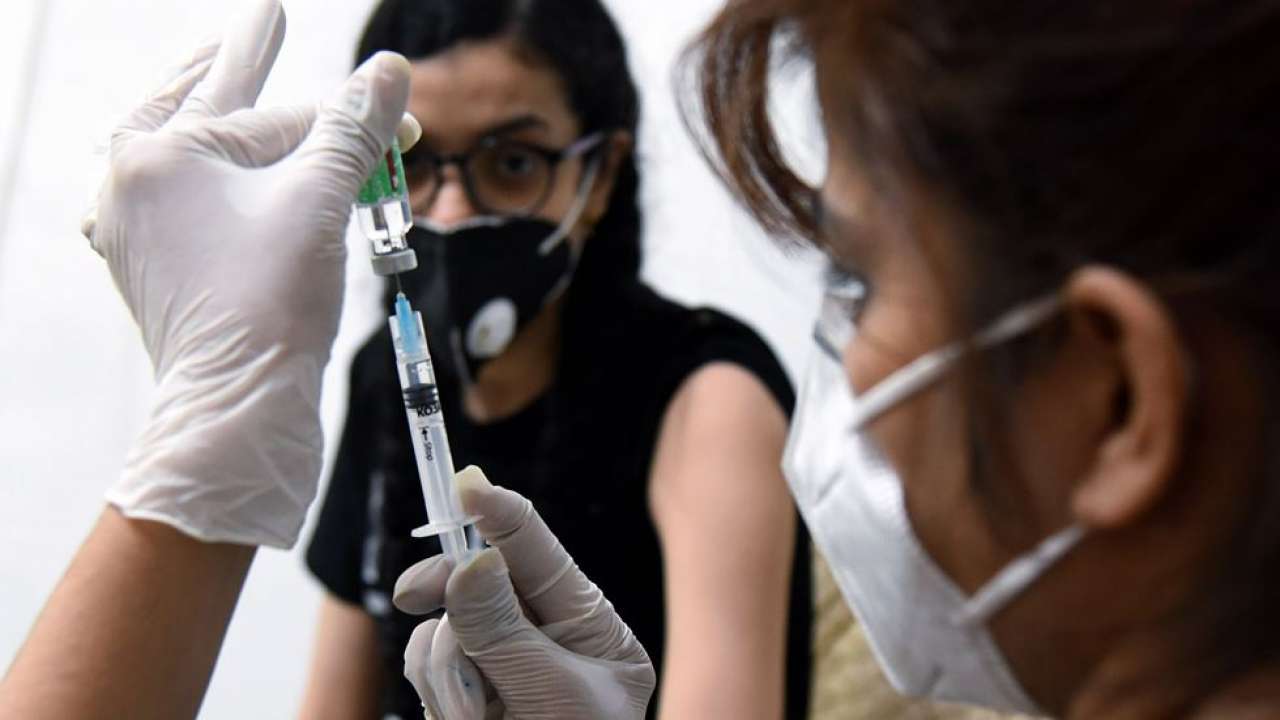
The trial run of the coronavirus vaccine for kids is underway in India. If the outcomes are in favour, then vaccination of kids would possibly begin from September-October, AIIMS Delhi Director Dr Randeep Guleria has mentioned.
In an interview with information company ANI, Dr Guleria mentioned that Bharat Biotech and different corporations are doing trials at a really quick tempo and hoped that information will result in approvals so that there’s a vaccine for kids within the nation by September-October. Covaxin’s information for kids after completion of section two or three trials is predicted by that point, he mentioned.
He famous that though youngsters often have a gentle illness, there’s a have to develop COVID-19 vaccines for them.
Covaxin is an indigenous vaccine developed by Bharat Biotech in collaboration with ICMR.
“Kids often have the gentle illness however we have to develop vaccines for kids as a result of if we’ve got to manage this pandemic everybody needs to be vaccinated. Bharat Biotech and different corporations are doing trials at a really quick tempo as dad and mom have come ahead with their youngsters for the trials,” he mentioned.
“One is hopeful that trial will likely be accomplished early and probably with comply with up of about two-three months, we will have information by September. Hopefully, by that point, approvals will likely be there in order that by September-October, we’ll have vaccines from our nation which we may give to youngsters,” he added.
Dr Guleria mentioned Bharat Biotech is conducting trials between the age group of two to 18 years and “this covers a large spectrum”.
Requested about Zydus Cadila vaccine, he mentioned it’s a DNA vaccine, a brand new platform is getting used and “it’s one thing that we should always pleased with”.
He added that it’s a platform for which analysis has not been completed in previous within the nation.
“The information remains to be being collated and one is hoping that they may have the ability to submit the information to the DCGI for regulatory approval. It would rely on the corporate how rapidly it is ready to collate the information and provides it to the regulatory authority,” he mentioned.
Requested about Pfizer CEO stating that the corporate is within the “remaining levels” of getting approval for its COVID-19 vaccine in India, Dr Guleria mentioned that the agency has been in negotiations for a while and these would have probably reached the ultimate stage.
“One is hopeful that it ought to happen as a result of the negotiations with the corporate is happening for fairly a while. I’m certain now they’re reaching a stage the place remaining type understanding will likely be there,” he mentioned.
(With ANI inputs)